Universal Solvent
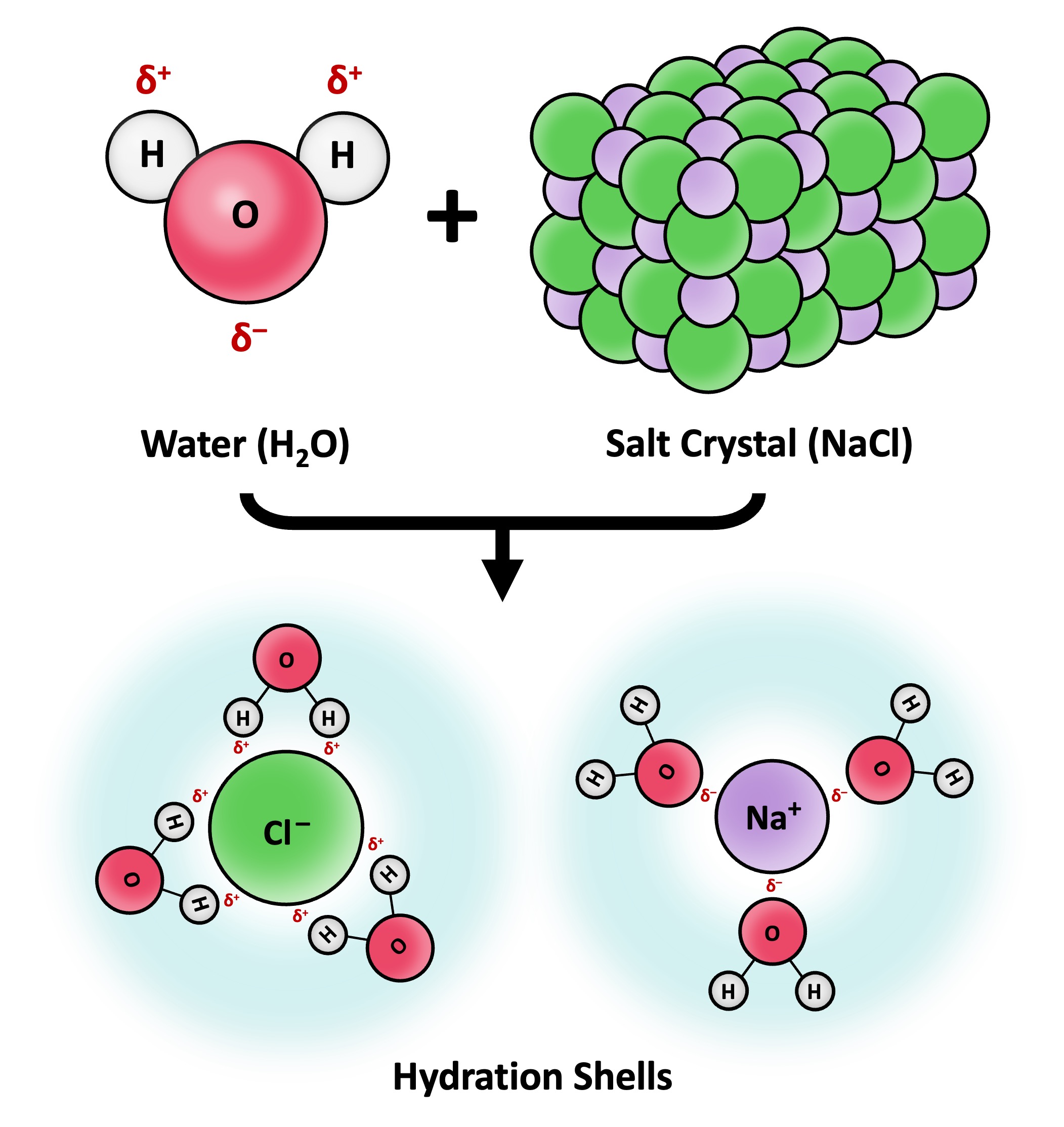
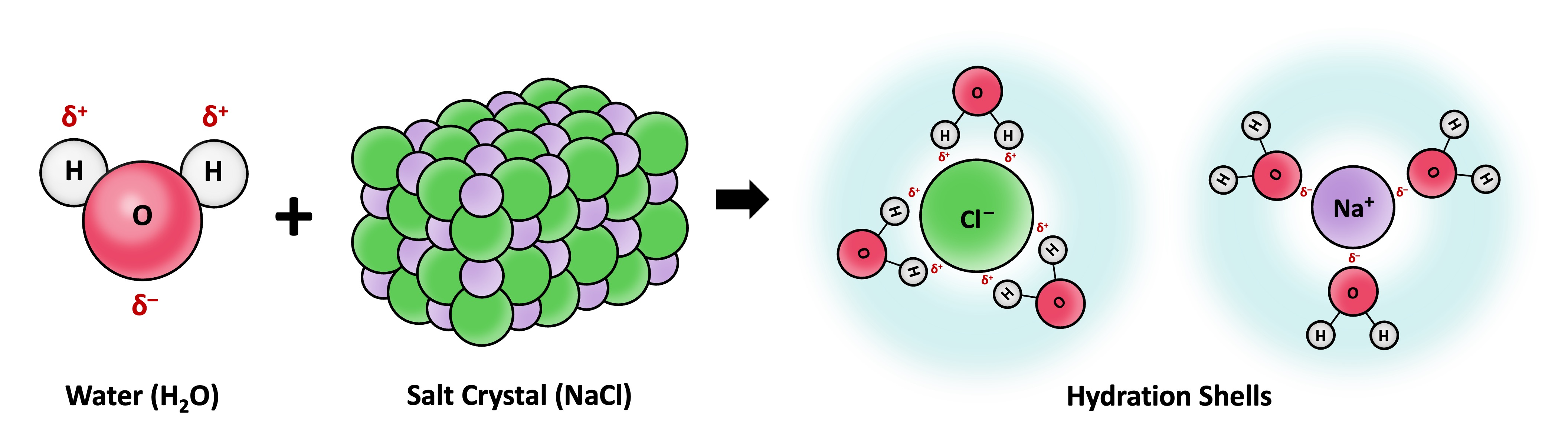
Solvation is the process by which solvent molecules surround and interact with solute molecules
-
A solute is any substance (usually a solid) that is dissolved by a solvent molecule to form a solution
-
Solutes that are able to be dissolved by a solvent are considered to be soluble (insoluble substances will not form a solution)
Water is commonly referred to as the universal solvent due to its capacity to dissolve a large number of substances
-
Water can dissolve any substance that contains charged particles (ions) or electronegative atoms (polarity)
-
The polar regions of the water molecule associate with molecular surfaces that have an opposing charge, forming dispersive hydration shells
Solvation