Fatty Acids
Fatty acids are long hydrocarbon chains that are found in most types of lipids (excluding steroids)
-
Fatty acids may differ in the length of the hydrocarbon chain, but most typically contain between 4 – 24 carbons
Types of Fatty Acids
Fatty acids can be classified according to the presence or absence of double bonds between the carbon atoms in the hydrocarbon chain
-
Saturated fatty acids possess straight hydrocarbon chains with no double bonds (they have the maximum possible number of H atoms)
-
Unsaturated fatty acids have double bonds – they can be either mono-unsaturated (1 double bond) or poly-unsaturated (>1 double bond)
Unsaturated fatty acids may occur in either of two distinct structural configurations – cis and trans isomers
-
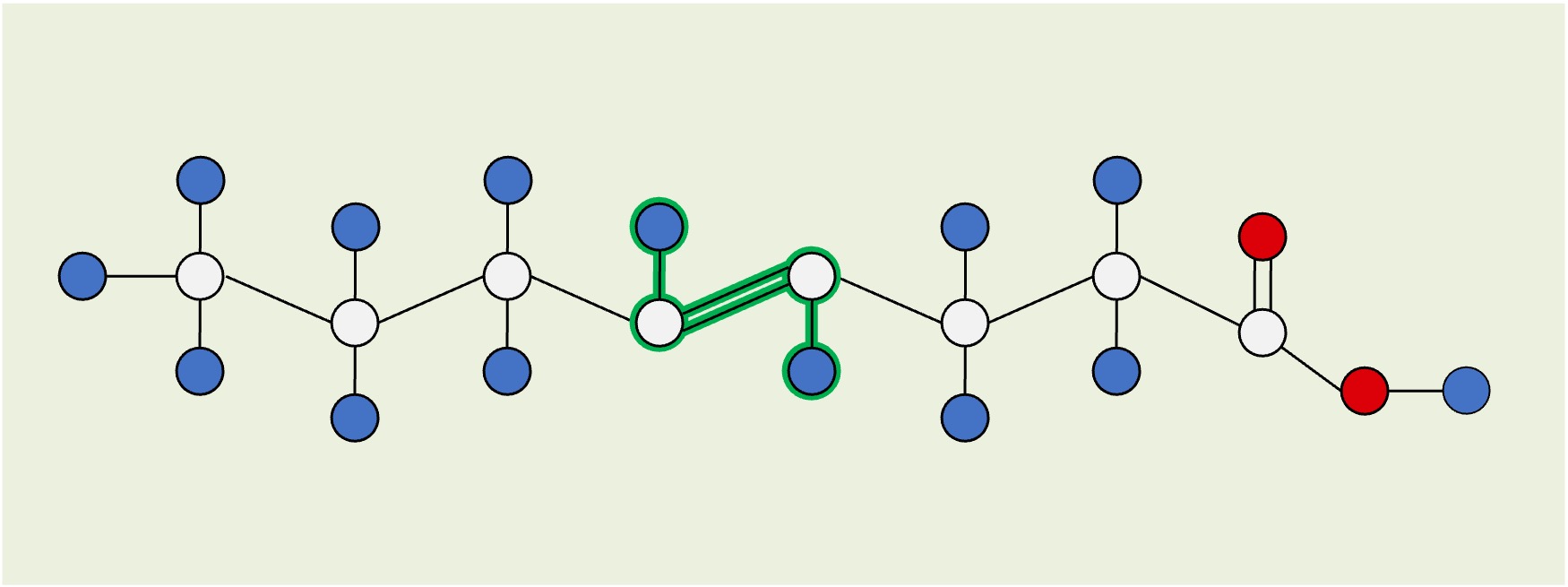
Cis: The hydrogen atoms attached to the carbon double bond are on the same side, causing the hydrocarbon chain to kink
-
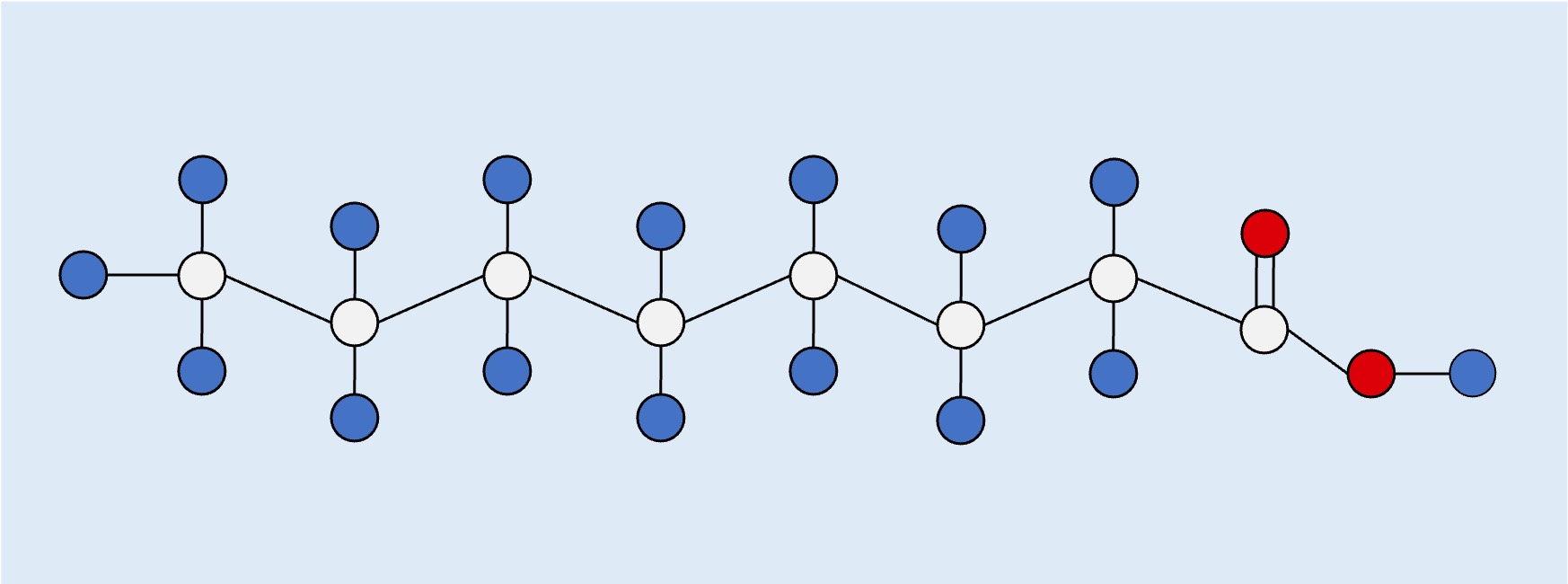
Trans: The hydrogen atoms attached to the carbon double bond are on different sides, meaning the hydrocarbon chain remains straight
In living organisms, unsaturated fatty acids are nearly always in the cis configuration – only ruminant animals (cows and sheep) produce trans fatty acids
Fatty Acid Structures
Saturated (no C=C double bonds)
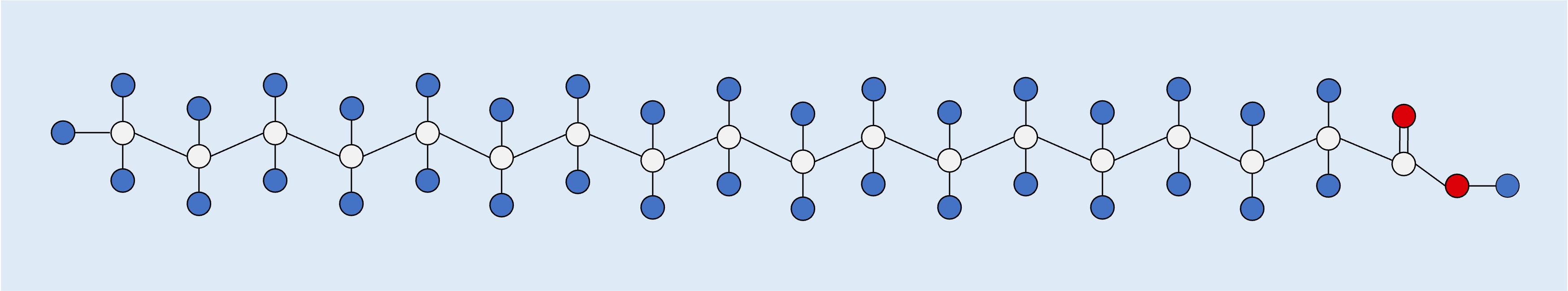
Saturated
Unsaturated (trans)

Unsaturated (cis)
Fats versus Oils
Living organism store their lipids as either fats or oils depending on the type of fatty acid involved (saturated or cis-unsaturated)
-
These fatty acids differ in the shape of their hydrocarbon chains (straight or bent)
Fats (Saturated)
-
Saturated fatty acids have straight chains that can be more tightly packed, making them more efficient for energy storage
-
However, this tight packaging also increasing the number of intermolecular forces between the fatty acid chains, resulting in a higher melting point
-
This means it takes higher temperatures to keep them liquid and they will typically exist as fats (solid at a room temperature of 25ºC)
Oils (Unsaturated)
-
Unsaturated (cis) fatty acids have kinked chains that cause them to be more loosely packed (poly-unsaturated fatty acids have multiple kinks and are consequently even more dispersed)
-
This means there are fewer intermolecular forces and less energy is required to separate the fatty acids, resulting in a lower melting point
-
Consequently, they will remain liquid at cooler temperatures and so usually exist as oils (liquid at a room temperature of 25ºC)
Fats vs Oil Comparison
-
Fats
Oils
Sources
-
Animals (endotherms)
-
Plants
Fatty Acids
-
Saturated
-
Unsaturated (cis)
-
-
Shape
-
Straight chains
-
Kinked chains
State
-
Solid at room temperature
-
Liquid at room temperature
Melting point
-
Comparatively high
-
Comparatively low
-
Note: Trans-fatty acids will share similar properties to saturated fatty acids (higher melting points and solid at room temperature) due to their hydrocarbon chains being straight
Lipid Storage
The storage of lipids as either fats or oils is primarily a consequence of an organism’s physiology and environment
-
Organisms need fatty acids that will remain liquid at their normal body temperature (to allow them to be utilized by the body)
-
However, organisms will try to produce the most tightly packed fatty acids possible in order to maximize their energy storage
Animals
-
Endotherms (warm blooded mammals) will tend to produce more saturated fats – as their higher core body temperatures can keep these fatty acids liquid
-
Conversely, ectotherms living in cold environments tend to produce cis-unsaturated oils – as saturated fats would solidify in the colder temperatures
-
This is why cold water fish oils are a rich source of poly-unsaturated fats that are essential to a human diet (omega-3 and omega-6)
Plants
-
As plants cannot control their own internal temperatures, they also predominantly produce cis-unsaturated liquid oils
-
However, tropical plants (found in warmer climates) will tend to produce comparatively more saturated fats than temperate plants (found in cooler climates)